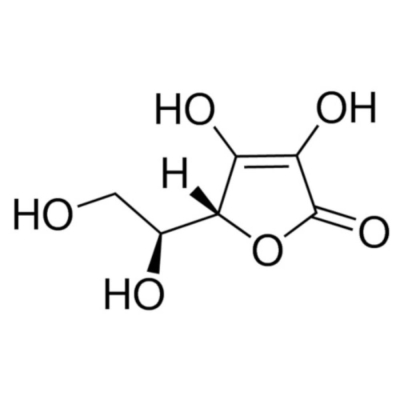
L-Ascorbic acid, Ph Eur
Biochem/physiol Actions
Ascorbic Acid, also known as Vitamin C, is a six-carbon lactone produced by plants and some animal species but not by humans and other primates.
L-Ascorbic acid functions according to its oxido-reduction property. It is a co-factor for hydroxylation. The biochemical actions of L-ascorbic acid is dependent on the activity of monoxygenase. It is involved in carnitine, collagen synthesis and production of neurotransmitters. Vitamin C exhibits anti-oxidant properties. Vitamin C plays a role in providing protection against photoaging and anti-aging effect. Deficiency of vitamin C leads to scurvy, bleeding gums, poor wound healing, anemia and muscle degeneration.
General description
L-Ascorbic acid is the most biologically active form of ascorbic acid. It is hydrophilic and an unstable compound. L-Ascorbic acid contains a six-carbon lactone produced by plants and some animal species but not by humans and other primates. Ascorbic acid, also referred to as Vitamin C, is a water-soluble vitamin. Ascorbic acid carries a neutral charge, which gets converted to ascorbate by protonation. Vitamin C is a part of citrus fruits, such as broccoli, strawberries, turnip.
| Colour | white to off-white |
|---|---|
| InChI key | CIWBSHSKHKDKBQ-JLAZNSOCSA-N |
| InChI | 1S/C6H8O6/c7-1-2(8)5-3(9)4(10)6(11)12-5/h2,5,7-10H,1H2/t2-,5+/m0/s1 |
| Manufacturer | SIGMA-ALDRICH |
| Agency | tested according to Ph. Eur., USP/NF |
| Form | solid |
| Assay | 99.0-100.5% |
There are no downloads for this product.